A dedicated Laboratory Information Management System, successfully implemented in PET Facilities around the world.
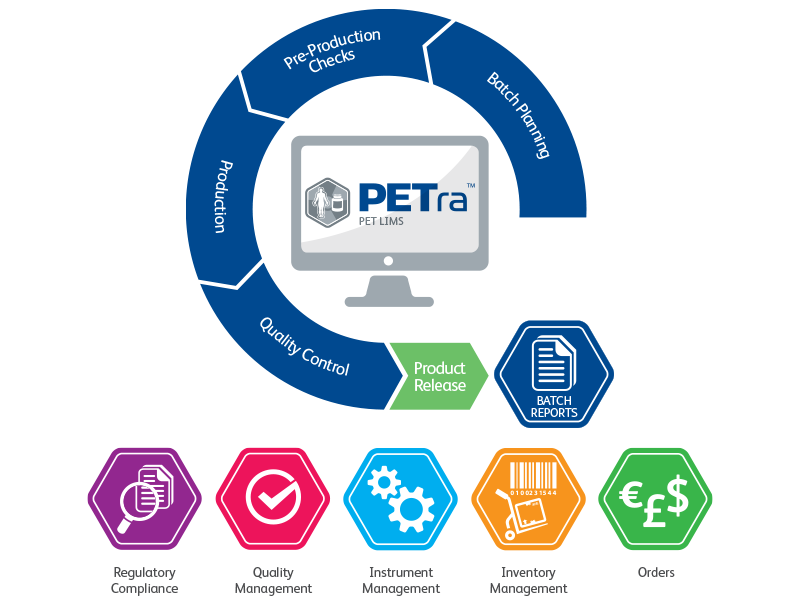
PETra is a true PET Laboratory Information Management System (LIMS), designed specifically for use within PET production facilities in order to improve efficiency and compliance.
Since it’s introduction to the market PETra has quickly become an industry favorite following successful installations in some of the world’s most prestigious and regulated PET facilities including Mallinckrodt Institute of Radiology (Washington University, St Louis, USA), Herlev Hospital PET Center (Copenhagen, Denmark), Memorial Sloan Kettering Cancer Research Center (New York, USA) and a multi-site deployment at Triad Isotopes (USA).
NEW: PETra PET LIMS six-part webinar series
View the webinar recordings from our recent webinar series below, including An Introduction to PETra, Batch Reporting, Inventory, QMS, Validation, and IT Requirements.
Direct Data Capture
During the various phases of PET production, facilities have to manage data from a range of equipment and software packages, resulting in multiple outputs and reports. What’s unique about PETra, is that it captures data directly from all equipment used in the process.
Eliminates Transcription Errors
As a result of direct data capture, PETra completely eliminates manual transcription and the likelihood of any errors.
Interfaces to all Equipment
PETra is capable of connecting with all the equipment used, either by interfacing directly with the instrument or connecting to the relevant software package. PETra is compatible with all leading manufacturers.
Improved Efficiency
Through managing data electronically PETra significantly improves workflow efficiency including:
- Automatic calculations
- Storing site, customer and dose information in one place
- Step-by-step prompts for pre-production checks
- Capturing data directly, eliminating transcription errors
- Batch reports ready at the click of a button without having to collate a range of reports
- Electronic signatures Not having to chase missing manual signatures
- Eliminating human errors on quality records
- Managing documents without a paper trail
- Built in procedures for all quality management tasks such as OOS, CAPA and Deviations
- Sending user and group notifications
- Having a secure and compliant system
- Trending any data
- Automatic inventory management using a barcoded system and notifications
Improved Compliance
LabLogic’s decades of experience and expertise in providing solutions within highly regulated environments means that PETra is an unrivalled solution for PET facilities.
Our system is built to ensure compliance with GMP, Pharmacopoeia and FDA regulations.
'PETra is a great solution for dealing with regulatory requirements specifically those from the FDA. My role as a responsible person is made so much easier when I can ensure all the data points have been collected, data is stored safely and I have instant access to all vital records'
Standardize Production
Multi-site facilities can standardize production of their radiopharmaceuticals with PETra.
Each site can be configured to ensure the same processes are being routinely carried out, removing inconsistencies.