The industry standard radiochromatography system used by thousands of scientists worldwide.
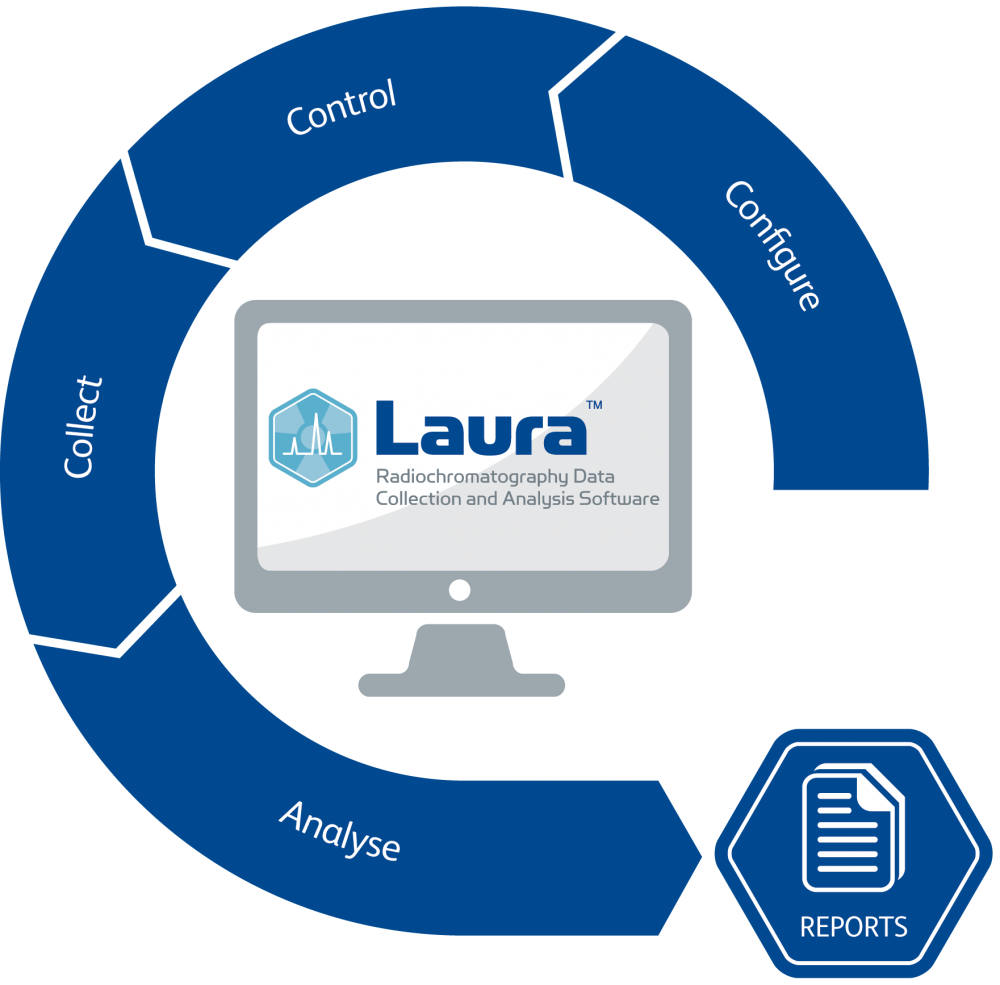
Laura is the ideal software package whether the work involves metabolite profiling, quality control, compound purification or any other chromatography task. It offers users the facility to create and edit methods, set up sample runs and view data collection across the network, in real time and without being confined to the bench-top PC.
Laura offers a Single Point of Control of both the radio-detector and HPLC system in a GxP environment; one system for all your radio-chromatography needs.
Introducing Laura 6
Laura 6 introduces brand new icons in an intuitive ribbon design, making access to Laura’s functionality quicker and easier than ever before. It also groups together logical functions, as well as providing improved tool tips, and contextual tabs to help the user find the function they require.
Functionality
Laura 6 offers full control of the radiochromatography process in a simple, easy to use format. Using a simple wizard driven process for multiple steps Laura is as intuitive to use for the occasional user, as it is for the researcher that uses Laura every day.
Laura 6 has all the features an experienced chromatographer expects - multiple trace display and data treatment, quench correction, decay correction, manual and automatic peak finding, scintillator pump control, fraction collector control, multiple run overlay.
Compliance
Laura 6 can be installed and configured to meet the most rigorous regulatory compliance. Featuring configurable audit trail, multi level security, e-signatures and data storage into a secure database environment Laura is the radiochromatographers answer for regulatory compliance.
Connectivity
Laura 6 provides connectivity to the widest range of detectors and chromatography systems. With a wide range of detectors supported including the B-RAM for radioactivity detection along with chromatography systems for HPLC, TLC, GC as well as multi channel analyzers for nuclide identification Laura is truly the data system for all your chromatography requirements.
To find an up to date and comprehensive list of all equipment Laura interfaces with, check out the Target Platform file from our downloads section.
Regulatory Compliance
Laura™ is designed to fully support GLP and associated regulatory compliance. This includes support for FDA 21 CFR part 11. Totally configurable to meet all requirements, Laura™ features audit trail, flexible security settings, multi-level access and e-signatures.
Electronic Signatures
Laura™ supports the use of electronic signatures and the system can be configured to suit the user sites requirements for electronic data.
Audit Trail
Laura™ has a fully featured audit trail that can again be configured to suit the requirements of the user site. Response from the user can be a simple as choosing from a pre-defined list of standard audit reasons or require a unique response depending on the situation and site rules.
Access Levels
Laura™ features multi-level access control and the system can be configured to have as many levels of access as the user site requires. Menu items are then restricted according to the security level of the individual logged in.
Security Settings
Laura™ can be configured to suit your particular site requirements for security. User names and passwords can be controlled relative to the site requirements as can inactivity time outs and all other functions required for a secure system.
Data Integrity
Regulatory compliance is an essential feature of Laura, built to meet GLP/GMP, MHRA and FDA 21 CFR part 212 / 11 requirements. Featuring configurable audit trail, electronic signatures, secure data storage in a database environment and multi-level security Laura
can be configured to meet regulatory requirements for data integrity.